Citation: PLOS One, 2017, doi: journal.pone.0179669
Abstract
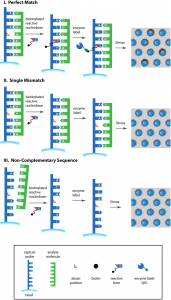
We have developed a single probe method for detecting microRNA from human serum using single molecule arrays, with sequence specificity down to a single base, and without the use of amplification by polymerases. An abasic peptide nucleic acid (PNA) probe—containing a reactive amine instead of a nucleotide at a specific position in the sequence—for detecting a microRNA was conjugated to superparamagnetic beads. These beads were incubated with a sample containing microRNA, a biotinylated reactive nucleobase—containing an aldehyde group—that was complementary to the missing base in the probe sequence, and a reducing agent. When a target molecule with an exact match in sequence hybridized to the capture probe, the reactive nucleobase was covalently attached to the backbone of the probe by a dynamic covalent chemical reaction. Single molecules of the biotin-labeled probe were then labeled with streptavidin-β-galactosidase (SβG), the beads were resuspended in a fluorogenic enzyme substrate, loaded into an array of femtoliter wells, and sealed with oil. The array was imaged fluorescently to determine which beads were associated with single enzymes, and the average number of enzymes per bead was determined. The assay had a limit of detection of 500 fM, approximately 500 times more sensitive than a corresponding analog bead-based assay, with target specificity down to a single base mis-match. This assay was used to measure microRNA-122 (miR-122)—an established biomarker of liver toxicity—extracted from the serum of patients who had acute liver injury due to acetaminophen, and control healthy patients. All patients with liver injury had higher levels of miR-122 in their serum compared to controls, and the concentrations measured correlated well with those determined using RT-qPCR. This approach allows rapid quantification of circulating microRNA with single-based specificity and a limit of quantification suitable for clinical use.